Shanghai, People’s Republic of China — Cook Medical has introduced the polymer-free Zilver® PTX® Drug-Eluting Peripheral Stent, complete with a user-friendly thumbwheel delivery system for single-handed deployment, in the Chinese Mainland. Cook is enthusiastic about providing this innovative, data-supported stent to physicians in the Chinese Mainland who specialize in treating peripheral vascular disease. This is one way Cook is delivering on our commitment to product leadership and making our industry-leading technology available in more areas.

The Zilver PTX Drug-Eluting Peripheral Stent was launched in the Chinese Mainland in 2020 and was intended for use in the treatment of symptomatic vascular disease of the above-the-knee femoropopliteal arteries having reference vessel diameter from 4 mm to 7 mm. Originally, the stent came with a pin-and-pull delivery system. Now, the new thumbwheel model of Zilver PTX Drug-Eluting Peripheral Stent has been approved for registration (Registration Number: 国械注进 20203130151) by China’s National Medical Products Administration.
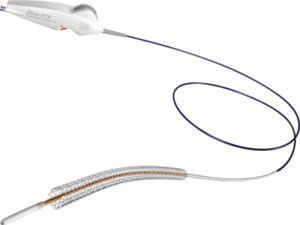
To use the device, a physician gains arterial access through the groin and guides a Zilver PTX stent to the narrowed artery with a catheter. The nitinol-alloy stent is deployed and expands like a scaffold to help keep the artery open after the catheter is withdrawn. It provides a support to treat lesion residual restenosis, and it reduces the elastic recoil after balloon dilatation. The stent surface is coated with 3μg/mm² paclitaxel, which inhibits the proliferation of vascular smooth muscle cells, and thereby is expected to reduce the occurrence of restenosis [1].The new thumbwheel delivery system includes an ergonomic handle with a safety lock and a Luer hub (flushing port). This allows for the release of the stent by slowly rotating the thumbwheel, making deployment simpler and more precise than the former pin-and-pull delivery system.
With the approval of the new thumbwheel delivery system, additional stent sizes are also available. Stent diameters range from 5 mm to 8 mm and stent lengths range from 40 mm to 140 mm. This broad range of stent sizes enables Cook to address a wider spectrum of patients’ clinical needs.
“We are excited to announce the availability of this product to healthcare professionals and patients in the Chinese Mainland. Since Cook established its first Chinese office in Beijing in 1996, we have valued our collaborations with physicians in tailoring solutions to meet their patients’ clinical needs. Now that Zilver PTX is available, physicians can have important conversations with patients about what kinds of stent options are available and what the long-term data may mean for a specific patient,” said Alec Cerchiari, Director of Product Management for Cook’s PAD & Venous specialty.
Beyond the product’s advantages, the Zilver PTX stent is supported by an extensive body of data proving its safety and effectiveness. Notably, Cook’s 5-year clinical data have shown that “Zilver PTX DES provided sustained safety and clinical durability compared to standard endovascular treatments[2].” Furthermore, Cook obtained approval for its drug eluting stent from China’s National Medical Products Administration only after conducting a clinical study in Chinese patients. The results of this study have been published [3].
For further insights into Zilver PTX, we invite you to explore our dedicated Zilver PTX resource page.
[1] Resource link: https://www.nmpa.gov.cn/yaowen/ypjgyw/ylqxyw/20200409143901774.html
[2] Dake MD, et al. Durable clinical effectiveness with paclitaxel-eluting stents in the femoropopliteal artery: 5-year results of the Zilver PTX randomized trial. Circulation. 2016;133:1472-1483. doi: 10.1161/CIRCULATIONAHA.115.016900.
[3] Ye W, et al. First peripheral drug-eluting stent clinical results from China: 1-year outcomes of the Zilver PTX China study. Front Cardiovasc Med. 2022;9:877578. doi: 10.3389/fcvm.2022.877578.
Disclaimer: 1. The summaries of the instructions contained herein are not intended to be a substitute for the product’s Instruction For Use (IFU). For more detailed information, including indications, contraindications, warnings and precautions, please refer to the IFU. 2. Please seek professional advice from a healthcare professional if you are involved in the diagnosis, treatment or rehabilitation of a disease. 3. The content of this article belongs to Cook Medical, and the related text and images have been authorized. Any individual or other media or websites wishing to reproduce this article must obtain prior authorization from Cook Medical and indicate the source of reproduction.
About Cook Medical
Since 1963, Cook Medical has worked closely with physicians to develop technologies that eliminate the need for open surgery. Today we invent, manufacture, and deliver a unique portfolio of medical devices to the healthcare systems of the world. Serving patients is a privilege and we demand the highest standards of quality ethics, and service. We have remained family owned so that we have the freedom to focus on what we care about: our patients, our employees, and our communities.
You can visit Cook’s global website at CookMedical.com and Cook China’s website at www.cookmedical.com.cn to learn more about our product and company news. You can also follow our official WeChat account by searching “CookMedical 库克医疗”.